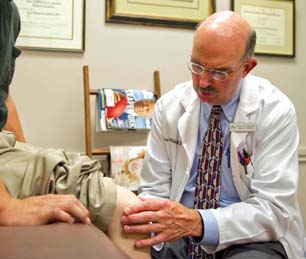
Investigator Initiated Research
AlloSource is dedicated to advancing the science of allografts. For this reason, as well as our commitment to our donors and our patients, we go above and beyond to gather evidence-based research to put safe, quality allografts into the hands of clinicians around the world.
Integrity is at the cornerstone of our clinical research program. AlloSource is currently conducting numerous studies both domestically and internationally, working closely with physicians and scientists across multiple orthopedic therapies to continuously improve and bring new allografts to the market, while simultaneously maximizing the gift of donation.
AlloSource’s commitment to improve and provide safe, quality tissue products to our clinicians is the cornerstone of our clinical research program. The AlloSource Investigator Initiated Research (IIR) Grant Program was established to support Investigator Initiated research that honors the gift of donation by maximizing its medical impact.
AlloSource accepts grant proposals for Investigator Initiated clinical research studies of AlloSource tissue and/or post-market product lines. All grant proposals are reviewed by the AlloSource Research Internal Review Committee (ARIRC) and, if approved, AlloSource will collaborate with the Investigator to help implement the proposed research project. Grant requests may include project funding and/or implantable tissue.
Submitting a Request
Please email Clinical Research to request a copy of our Investigator Initiated Research Request Form at clinicalresearch@allosource.org.
Review Process
The AlloSource Research Internal Review Committee (ARIRC) meets on a regular basis to review grant proposals. Grand decisions are based on scientific, medical and/or scholarly significance, as well as alignment with corporate research priorities and available resources. All grant requestors are notified of the committee’s decision, or the status of their submission, after each meeting.
Upon approval of a request, AlloSource will collaborate with the Investigator/site, as a resource, to strengthen the scientific merit of the subsequent study documents and to ensure final study documents reflect the approved proposal.
Requirements for IIR
AlloSource requires the following completed documentation before an approved study can commence:
- Fully executed Research Agreement
- IRB/Ethics Committee approval
- IRB approved informed consent form (ICF) (if applicable)
- Final study protocol
- Final case report forms (CRFs)/data collection spreadsheets
- Quality of Life assessment templates (if applicable)
- AlloSource approved Adverse Event/Serious Adverse Event Form
- Curricula Vitae of Investigator(s) (Renewed every 2 years or as necessary by date of expiration)
- Current State Medical License of Investigator(s) (Renewed annually or as necessary by date of expiration)
- Delegation of Authority Log
- Proof of ClinicalTrials.gov acceptance and NCT Number
- IRS W9 Form
Study Maintenance
AlloSource requires study status updates. Frequency is determined based on study design and complexity. Updates are expected to include information on enrollment (if applicable), study milestones and/or completion dates and white paper/publication status (as appropriate).
- AlloSource requires notification, and rights of approval, of any proposed amendment to the approved protocol, ICF and CRF’s after the research has begun.
- AlloSource reserves the option to include the right to audit sites, generate data queries and asses that all applicable FDA regulations and ICH GCP Guidelines are being met.
Study Closure
In accordance with AlloSource’s Research Sponsorship Agreement, upon study completion Investigators are required to provide the following information:
- Certification that the study was conducted per the study design
- Written report, or raw data, of the final study results
- Draft of white paper/article proposal in advance of submission to publication
- Documentation/proof that grant funds and/or tissue were used solely to conduct the study as designed
- Confirmation of safety reporting
Publishing Results
In accordance with AlloSource’s Research Agreement, the Principal Investigator must submit a draft of the white paper/article proposal in advance of submission to a publication or publishing to the Clinical Research Department. Submissions must comply with all International Committee of Medical Journal Editors (ICMJE) recommendations (“The Uniform Requirements”), www.icmje.org.
If you are interested in partnering with AlloSource on clinical research, please contact Clinical Research at clinicalresearch@allosource.org
or 720.873.4811.